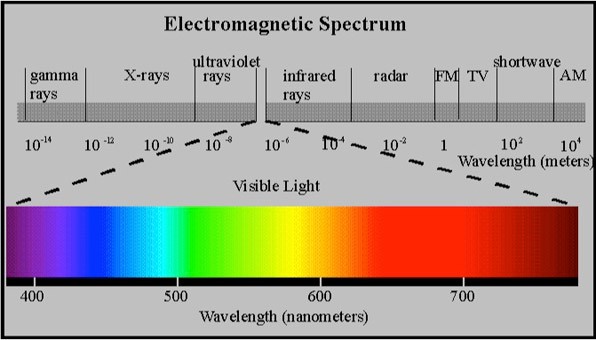
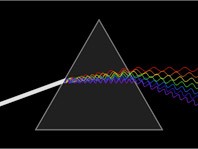
This well-known diagram illustrates an image that shows the electromagnetic spectrum with the visible wavelength expanded showing the section from 400 – 750 nm. This section is the portion of energy that can be perceived by receptors in eyes and processed by the brain. It provides human beings with the ability to see color.
Chemical Characterization of Pigments in Art
What Is Color?
"Color is the visual perceptual property corresponding in humans to the categories called red, green, blue, and others. Color derives from the spectrum of light (distribution of light power versus wavelength) interacting in the eye with the spectral sensitivities of the light receptors. Color categories and physical specification of color are also associated with objects, materials, lights sources, etc., based on their physical properties such as light absorption, reflection, or emission spectra. By defining a color space, colors can be identified numerically be their coordinates" (Wikipedia). Color is not a tangible thing. It is a perception.

Giovanni Bellini and Titian, The Feast of the Gods, 1514/1529, oil on canvas, National Gallery of Art, Widener Collection, 1942.9.1
Why does light appear to be “colorless?” White light can be dispersed by a prism because the relative speed of different wavelengths changes in a material such as glass. When all the wavelengths arrive at our eye, we perceive no color, just white. However, if an object is made from molecules that absorb blue and green light, then we will perceive that object as red.
This is how color is understood by artists. It is a material entity and not thought of in terms of wavelength at all.
Artists' colorants come from many sources: animal, mineral, vegetable, and synthetic.
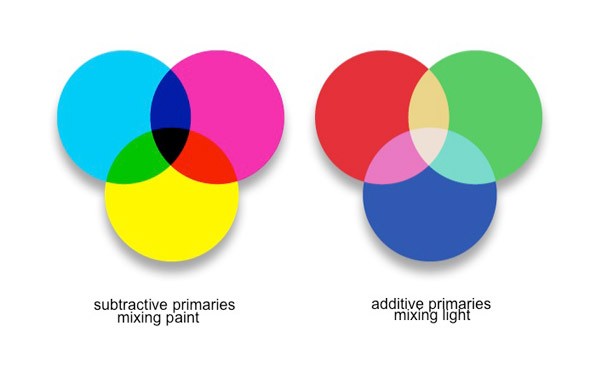
The artist lives in the material world where yellow and blue make green, because those pigments mixed together create that hue. Scientists more often work in the red, blue, and green world of additive color. However, additive color from a perceptual viewpoint appears to contradict what people see in the world around them.
Some Sources of Color
| Physical | Chemical |
|
|
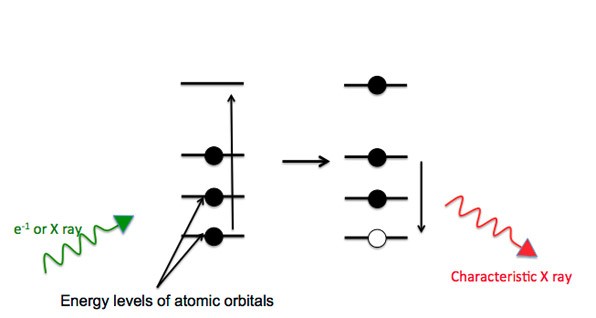
Color can be created by physical and chemical occurrence. Most chemical interactions that explain why materials have color are the result of electronic transitions. For example, they can be atom-centered, such as ligand field transitions in transition metal ions (resulting in the blue color of hydrated copper sulfate), or they can be intramolecular.
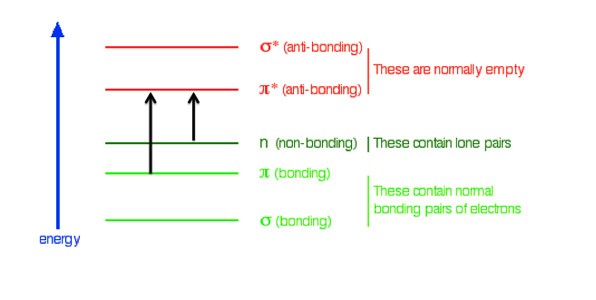
In some organic compounds with delocalized electrons the energy difference between the bonding and non-bonding pi and n orbitals and the anti-bonding pi star orbitals is in the visible region of the electromagnetic spectrum. When some photons are absorbed from "white light," we perceive color.
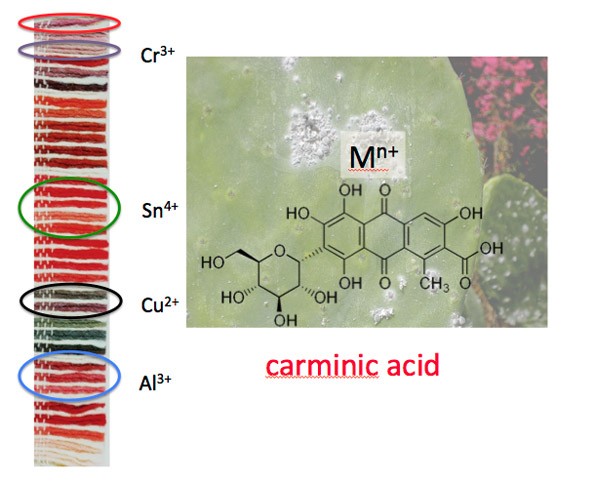
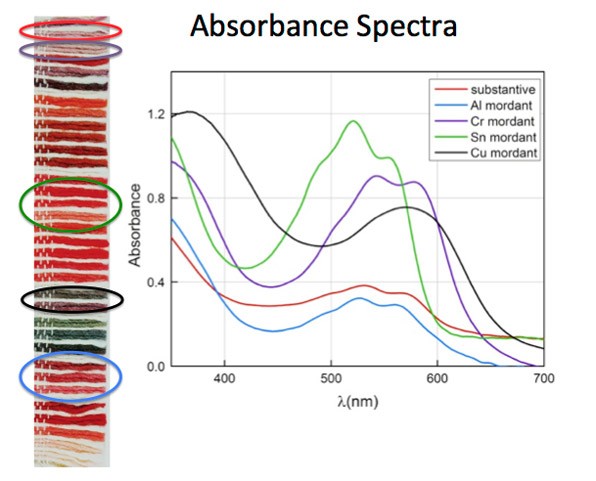
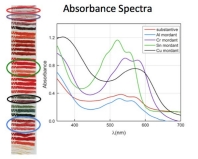
Carmine, a red colorant derived from cochineal insects, when studied scientifically, reveals that a variety of different shades of red—some that appear violet and others that are bright pink—can be fabricated by using different chemicals, called mordants, to process the raw dye material extracted from the cochineal insects.
This graph shows the absorbance spectral curves of cochineal dye based on the type of chemical mordant used to prepare them.
Some of the Analytical Methods Used to Identify Colorants in Works of Art
| Sample required | No Sample Required |
|
|
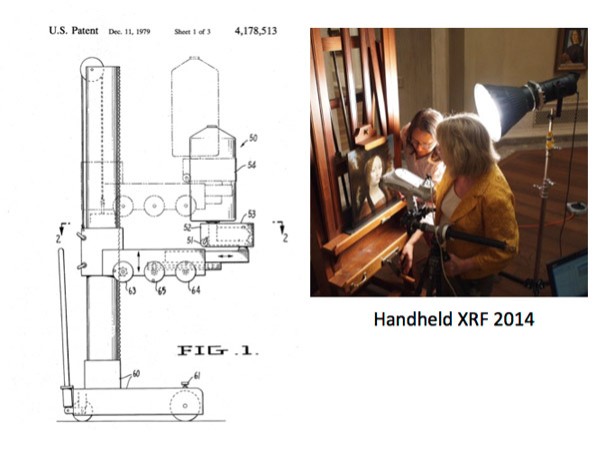
Illustrated are two scientists using a handheld X-ray fluorescence device to determine the chemical makeup of a pigment contained in a painting.
One method of capturing information from a sample is X-ray fluorescence; another is energy dispersive X-ray analysis.
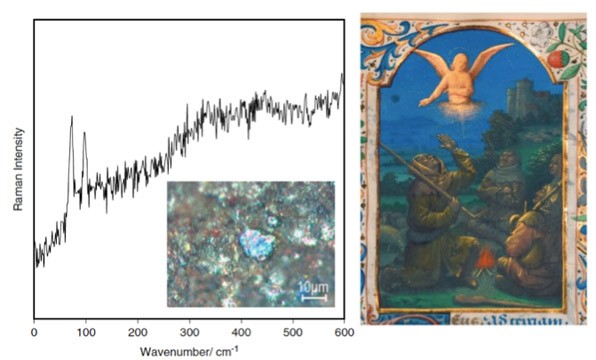
Several other analytical instruments can be used to obtain information about the chemical nature of pigments in works of art—these include Fourier transform infrared spectroscopy, near-infrared imaging, and Raman spectroscopy.
A surprising result found by a colleague in the field of conservation science, Karen Trentleman, indicates the artist's use of metallic bismuth to make a gray paint! Micro-Raman was used to examine a single particle and the spectra showed that metal was used and not an oxide or a sulfide.
Other nondestructive methods of collecting valuable research data include the instruments illustrated here. These camera-like devices capture information that a detector can analyze to determine the composition of a pigment used by an artist. In some instances, very minute samples are taken from existing cracks or the edges of a painting. Amazingly, a vast amount of information can be obtained from a tiny sample, smaller than a head of a pin.
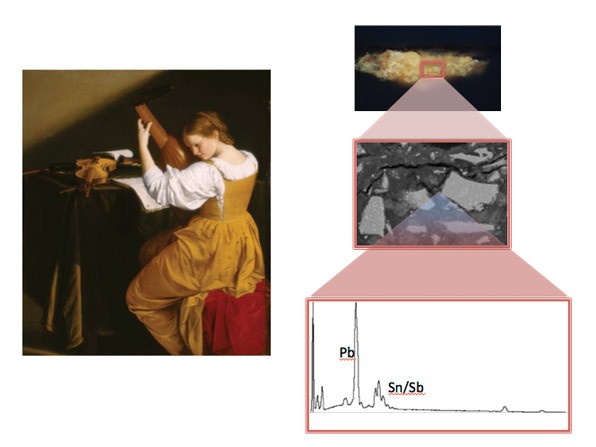
The sample comes from a loss. A scientist mounts a sample in a plastic block. Most samples are very small—but when placed under a microscope and photographed, the sample can be seen rather easily. When scientists look at the sample using a microscope, they can identify all the layers of the painting. In this case, there is a white chalk ground, a line of drawing, and two or three layers of paint that blend together, each mixed from lead white and azurite—indicating that the artist worked from light to dark.
Scientists can examine the paint sample using scanning electron microscopy along with energy dispersive X-ray analysis to identify and characterize the inorganic colorants.
The sample taken from Gentileschi’s painting The Lute Player, c. 1612/1620, shows a small rectangle magnified by a microscope in the images on the top right. A portion of that area is examined using the scanning electron microscope, which yields the image in the center right. The energy dispersive X-ray instrument that is attached to the scanning electron microscope reveals the presence of lead (Pb) along with antimony and tin (Sn/Sb), which indicates that the golden brown pigment in The Lute Player is lead-tin antimony yellow.
References
“Color.” Wikipedia: The Free Encyclopedia. Wikimedia Foundation, Inc. (accessed October 15, 2015), https://en.wikipedia.org/wiki/Color.
"Scientific Research Department Publications." National Gallery of Art (accessed October 15, 2015), http://www.nga.gov/conservation/publications/publication-list.html.
Sgamellotti, A., B.G. Brunetti, and C. Miliani. Science and Art: The Painted Surface. Royal Society Of Chemistry, 2014.
Berrie, B.H. Rethinking the History of Artists' Pigments through Chemical Analysis. Annual Review of Analytical Chemistry (2012), 5: 441–459, http://www.annualreviews.org/doi/abs/10.1146/annurev-anchem-062011-143039.
Casadio, Francesca, Alyson V. Whitney, and Richard P. Van Duyne. "Plasmonic Nanostructures for Highly Sensitive Detection of Traditional Artists' Red Dyestuffs with Surface Enhanced Raman Spectroscoph." The Art Institute of Chicago (accessed October 15, 2015), http://www.eu-artech.org/files/Ext_ab/Casadio.pdf.
Shugar, A.N., and J.L. Mass. Handheld XRF for Art and Archaeology. Leuven University Press, 2012.
Serrano A., A. van den Doel, M. van Bommel, J. Hallett, I. Joosten, and K.J. van den Berg. Investigation of Crimson-Dyed Fibres for a New Approach on the Characterization of Cochineal and Kermes Dyes in Historical Textiles. Analytica Chimica Acta, http://dx.doi.org/10.1016/j.aca.2015.09.046.
"Note on Color Fading:'Paintings Fade Like Flowers.'" The Metropolitan Museum of Art (accessed October 15, 2015), http://www.metmuseum.org/exhibitions/listings/2015/van-gogh/color-fading.
Conservation Division
National Gallery of Art
2000B South Club Drive
Landover, MD 20785
[email protected]